Abstract
Background
Chimeric antigen receptor (CAR)-T cell therapy has revolutionised the treatment of relapsed or refractory B-ALL in children and young adults with unprecedented response rates. However, primary resistance and relapse are unresolved challenges that limit long term benefit in a significant proportion of patients. Death receptor mediated extrinsic apoptosis is a key component of CAR-T cytotoxicity and impairment of this system, of which CAR-T derived TRAIL (tumour necrosis factor related apoptosis inducing ligand) is a key initiator, is a principal driver of primary resistance. Arginine deprivation with the therapeutic enzyme ADI-PEG20 (pegylated arginine deiminase) sensitises cancers deficient in the enzyme argininosuccinate synthase (ASS1) to the apoptosis initiating activity of TRAIL through tumour cell surface upregulation of death receptors DR4 and DR5. Whether this effect could potentiate the TRAIL-DR activity in CAR-T therapy has not been explored.
Aim
We tested the hypothesis that ADI-PEG20 treatment can sensitise susceptible B-ALL to anti-CD19 CAR-T through priming of death receptor mediated apoptosis signalling.
Methods
The effect of ADI-PEG20 on cell survival and death receptor expression in B-ALL cell lines and primary samples was analysed by flow cytometry. Second generation anti-CD19 CAR-T cells with a CD28 costimulatory domain were generated by retroviral transduction of activated peripheral blood mononuclear cells (PBMC) from healthy donors. For CAR-T co-culture experiments, B-ALL cell lines were pre-treated with ADI-PEG20 before washing and re-suspending in arginine replete media prior to CAR-T cell addition.
Results
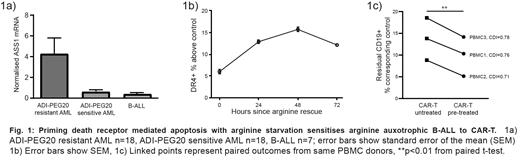
To establish potential susceptibility of B-ALL to ADI-PEG20 we measured expression of ASS1, which inversely correlates with sensitivity to the drug, using combined in situ immunohistochemistry (n=6) and RT-qPCR (n=7). ASS1 deficiency was consistently seen in this series of primary samples suggesting the potential utility of ADI-PEG20 in B-ALL, with comparable expression levels to those seen in a cohort of primary AML samples proven to be sensitive to the drug (figure 1a).
Next, to examine variation in ASS1 expression between genetically defined subtypes of B-ALL we re-analysed transcriptome data from a cohort of 215 patients treated on the ECOG E2993 trial, filtered into a network of 58 genes generated according to known or predicted interaction with ASS1. We found an enrichment of Philadelphia chromosome positive (Ph+) and Philadelphia-like (Ph-L) samples in the cluster characterised by lowest ASS1 expression along with high HIF1A expression, matching a recurrent pattern reported in other ADI-PEG20 sensitive tumours. This therefore predicts that among B-ALL subtypes, Ph+ and Ph-L are likely to be most sensitive to therapeutic arginine deprivation.
We then functionally confirmed, using in vitro cell line (n=3) and in vivo patient derived xenograft models of B-ALL (n=2), that ASS1 deficiency is required for ADI-PEG20 sensitivity. Using the ASS1-low, Ph-L cell line MUTZ-5, we established that ADI-PEG20 induced apoptosis accompanies cell surface upregulation of both DR4 and DR5 expression. Upregulation of DR4 was observed to follow an upwards trend after treated cells were washed and re-suspended in arginine replete media, suggesting that transient arginine starvation can commit ASS1-low B-ALL to a state of apoptotic priming (figure 1b).
With confirmed engagement of arginine starvation and death receptor upregulation we tested the synergy potential of ADI-PEG20 pre-treatment of MUTZ-5 followed by CAR-T, utilising calculated combination drug indices (CDIs). Across independent PBMC donors (n=3) we observed greater potency killing of CD19 + leukaemia cells in the combination treated co-cultures when compared to the single agent treated conditions, with CDIs consistently less than 1 confirming a synergistic effect (figure 1c).
Conclusion
Our study proposes a synergistic interaction between the arginine depleting enzyme ADI-PEG20 and anti-CD19 CAR-T for the treatment of ASS1 deficient B-ALL, whereby priming of death receptor signalling may underlie enhanced CAR-T cytotoxicity against CD19 + tumour cells. These data support an emerging framework for CAR-T optimisation based on targeting of the death receptor mediated extrinsic apoptosis pathway and can inform future refinements in the development of cellular immunotherapy.
Bomalaski: Polaris Pharmaceuticals Inc.: Current Employment. Maher: Leucid Bio: Other: Chief Scientific Officer. Gribben: Abbvie: Honoraria; AZ: Honoraria, Research Funding; BMS: Honoraria; Gilead/Kite: Honoraria; Janssen: Honoraria, Research Funding; Morphosys: Honoraria; Novartis: Honoraria; Takeda: Honoraria; TG Therapeutis: Honoraria.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal